| 種由来 |
Tetrahymena
|
| 由来詳細 |
[Species]Tetrahymena thermophila
|
| 適用 |
Immuno Fluorescence
Immunocytochemistry (cell)
|
| 免疫動物 |
Mouse
|
| クローン |
21A10
|
|
※サムネイル画像をクリックすると拡大画像が表示されます。
Fig.1 Immunofluorescene staining of Nup98 in HeLa cells using 13C2 , 21A10 , or 2H10 monoclonal antibodies. Black-and-white images were obtained with mAb and DAPI. Color images represent merged images of mAb (green) with DAPI (magenta).
HeLa cells were cultured in a glass-bottom dish (MatTek, Ashland, MA) for 2 days before fixation. The cells were fixed with cold methanol (-30°C) for 30 min. After washing with PBS, all fixed samples were blocked with 1% BSA for 2 hr at room temperature. mAbs 13C2 and 21A10 were diluted to 0.5 μg/ml in PBS. Anti-Nup98 rat mAb 2H10 was also diluted to 0.5 μg/ml in PBS. Blocked samples were treated with these primary antibody solutions overnight at 4°C. The secondary antibodies were 4 μg/ml of Alexa488-labelled anti-mouse IgG for 13C2 and 21A10 or anti-rat IgG for 2H10. DNA was stained with 4’,6-diamidino-2-phenylindole (DAPI). Samples were washed three times with PBS between treatments. Fluorescence images were obtained with an Olympus fluorescence microscope IX-70 controlled by DeltaVision microscope system (Applied Precision, Issaquah, WA).
Both 13C2 and 21A10 mAbs stained the nuclear periphery of the human cells in a punctate pattern similar to that of 2H10 mAb. The signal at the nuclear periphery with 21A10 mAb was much higher and the background lower than that with 2H10 and 13C2 antibosies.
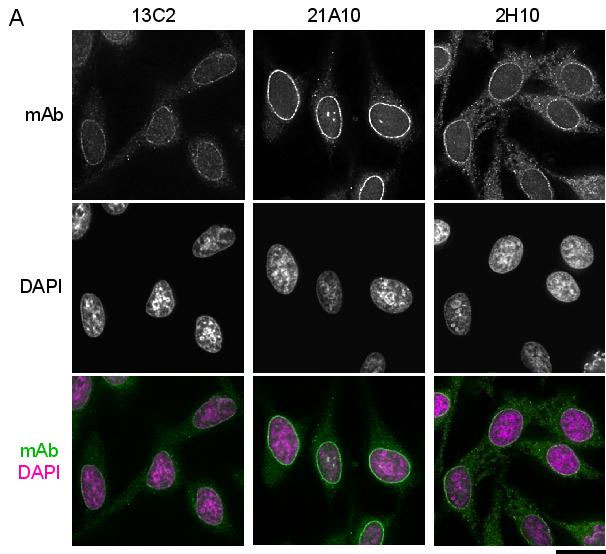
Fig. 2. Western blot analysis of Nup98 in HeLa cells with anti-Nup98 antibodies.
mAbs used are indicated on the tops of the lanes. Signals were detected using the same staining conditions, except that the exposure time of the middle lane (labeled 21A10 long exposure) was about 10 times longer than the exposure times of the other lanes. Open arrowheads represent the positions of Nup98. mAB 13C2 is the best for WB among them.
After SDS-PAGE, proteins were transferred to polyvinylidene difluoride (PVDF) membranes at 0.8 mA/cm2 for 2 hr using a semi-dry blotting apparatus. Membranes were blocked with 5% skim milk in PBS containing 0.1% Tween20 (PBS-T) for 3 hr at room temperature. The membranes were then incubated with mAbs diluted to 0.4 μg/ml in PBS containing 1% skim milk overnight at 4°C.. Membranes were then washed three times for 10 min with PBS-T followed by incubation with 0.4 μg/ml horseradish peroxidase (HRP)-labeled anti-mouse IgG or anti-rat IgG for 13C2 and 21A10 or 2H10, respectively, for 2 hr at room temperature. The membranes were then incubated with ImmunoStar reagent (Wako) for 1 min, and chemiluminescence signals were detected by an image analyzer.
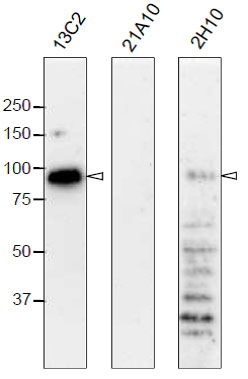
Fig. 3. Immunofluorescene staining of Nup98 in Tetrahymena themophila cells using 13C2 , 21A10 , 2H10 or 414 monoclonal antibodies. Black-and-white images were obtained with mAb and DAPI. Color images represent merged images of mAb (green) with DAPI (magenta). Dotted lines represent the outlines of cells. The open arrow indicates the micronucleus. Insets are magnified images showing the position of the micronucleus. Bar, 20 μm.
The cells were fixed with cold methanol (-30°C) for 30 min. After washing with PBS, all fixed samples were blocked with 1% BSA for 2 hr at room temperature. mAbs were diluted to 0.5 μg/ml in PBS. Blocked samples were treated with these primary antibody solutions overnight at 4°C. The secondary antibodies were 4 μg/ml of Alexa488-labelled anti-mouse IgG for 13C2, 21A10 and 414 or anti-rat IgG for 2H10. DNA was stained with 4’,6-diamidino-2-phenylindole (DAPI). Samples were washed three times with PBS between treatments.
Both 13C2 and 21A10 mAbs stained the macronuclear periphery of T. thermophila. 13C2 mAb was highly specific to the macronucleus (see 13C2 "mAb" inset). In contrast, in addition to clear macronuclear staining, 21A10 mAb also stained the micronuclear periphery (see 21A10 "mAb" inset). This indicates that 21A10 mAb recognizes Nups localizing to the micronucleus such as Nup308 in addition to MacNup98A. Neither mABs 2H10 nor 414 could stain nuclear periphery of Tetrahymena.
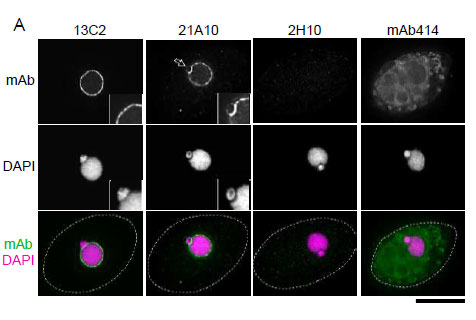
Fig. 4. Detection of MacNup98A of Tetrahymena by Western blotting with monoclonal antibodies 13C2 and 21A10. WB analysis of T. thermophila specimen from wild type (left 4 lanes) or GFP-MacNup98A expressing cells (right most lane). mAbs used are indicated on the tops of the lanes. Signals were detected using the same staining conditions, except that the exposure time of the middle lane (labeled 21A10 long exposure) was about 10 times longer than the exposure times of the other lanes. Open arrowheads and the closed arrow represent the positions of MacNup98A and GFP-MacNup98A, respectively. Diamonds and asterisks represent uncharacterized proteins.
Both 13C2 and 21A10 mAbs detected a protein with an apparent molecular weight (MW) of 98 kDa. This value approximates the MW of MacNup98A (112 kDa) deduced from the DNA sequence. Ectopically expressing GFP-MacNup98A in addition to endogenous MacNup98A (see right most lane, labeled GFP-MacNup98A) were detected by 13C2, with MWs of 98 and 125 kDa in the samples (arrows in “GFP-MacNup98A”). The 125-kDa band corresponds to GFP-MacNup98A, and is approximately 28 kDa larger than the endogenous protein detected by 13C2. As the MW of GFP is 28 kDa, suggesting that the 98-kDa protein band is endogenous MacNup98A. 2H10 mAb, also detected a protein with the same MW as endogenous Nup98A, but the band was very faint. For detection of MacNup98A of Tettrahymena with Western blotting, 13C2 is the best among the three antibodies.
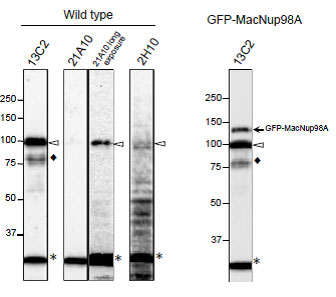
Fig. 5. Immunofluorescene staining of Nup98 in S. pombe cells with 13C2 , 21A10, or 2H10 monoclonal antibodies. Black-and-white images were obtained with mAb and DAPI. Color images represent merged images of mAb (green) with DAPI (magenta). Dotted lines represent the outlines of cells.Bar, 5 μm. S. pombe cells were fixed with 4% formaldehyde for 10 min, treated with 0.6 mg/ml Zymolyase 100T at 36°C for 70 min, and permeabilized with 1% Triton X-100 for 1 min. 1/10 dilutions of the supernatants of hybridoma cultures of 13C2 or 21A10 or 10 μg/ml IgG solution of 2H10 were used. Both 13C2 and 21A10 mAbs stained the nuclear periphery of S. pombe in a punctate pattern similar to that of the 2H10 mAb

Fig. 6. Detection of Nup98 in S. pombe cell extracts by Western blotting with monoclonal antibodies 13C2, 21A10 and 2H10. 1/10 dilutions of the supernatants of hybridoma culture medium of 13C2 or 21A10 or 1 μg/ml IgG solution of 2H10 were used. Left and right lanes represent specimens from a wild type strain and an S. pombe strain in which Nup98 was chromosomally replaced with Nup98-GFP, respectively. S. pombe cells harvested were heated in distilled water at 95°C for 5 min, suspended in 100 mM phosphate buffer (pH 6.8), 4 M urea, 2.5% SDS, 0.05 mM EDTA, and disrupted by glass beads using a Multi Beads Shocker (Yasui Kikai, Osaka,). The sample was clarified by centrifugation, and the supernatant was used as the whole cell extract. WB was performed as described above. 13C2 antibody among the three gave best result for WB of Nup98 in S. pombe.
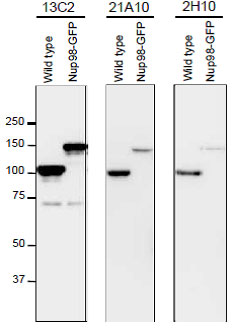
Fig. 7. Monoclonal antibodies 13C2 and 21A10 crossreact with multiple nucleoporins of S. cerevisiae.
(A) IF staining of S. cereviciae cells using 13C2, 21A10, or 2H10 mAbs: 1/10 dilutions of the supernatants of hybridoma cultures of 13C2 or 21A10 or 10 μg/ml IgG solution of 2H10 were used. Black-and-white images represent fluorescence images obtained with mAb (top) and DAPI (middle). Color images represent merged images of mAb (green) with DAPI (magenta). Dotted lines represent the outlines of cells. Bar, 5 μm. Methods were as described for S. pombe.
(B) WB analysis of S. cereviciae cell extract using 13C2 , 21A10, or 2H10 mAbs: 1/10 dilutions of the supernatants of hybridoma culture medium of 13C2 or 21A10 or 2 μg/ml IgG solution of 2H10 were used. Arrows represent the positions of the indicated nucleoporins. Asterisks represent uncharacterized proteins.
Methods were as described for S. pombe.

|

|
|
Fig.1 Immunofluorescene staining of Nup98 in HeLa cells using 13C2 , 21A10 , or 2H10 monoclonal antibodies. Black-and-white images were obtained with mAb and DAPI. Color images represent merged images of mAb (green) with DAPI (magenta).
HeLa cells were cultured in a glass-bottom dish (MatTek, Ashland, MA) for 2 days before fixation. The cells were fixed with cold methanol (-30°C) for 30 min. After washing with PBS, all fixed samples were blocked with 1% BSA for 2 hr at room temperature. mAbs 13C2 and 21A10 were diluted to 0.5 μg/ml in PBS. Anti-Nup98 rat mAb 2H10 was also diluted to 0.5 μg/ml in PBS. Blocked samples were treated with these primary antibody solutions overnight at 4°C. The secondary antibodies were 4 μg/ml of Alexa488-labelled anti-mouse IgG for 13C2 and 21A10 or anti-rat IgG for 2H10. DNA was stained with 4’,6-diamidino-2-phenylindole (DAPI). Samples were washed three times with PBS between treatments. Fluorescence images were obtained with an Olympus fluorescence microscope IX-70 controlled by DeltaVision microscope system (Applied Precision, Issaquah, WA).
Both 13C2 and 21A10 mAbs stained the nuclear periphery of the human cells in a punctate pattern similar to that of 2H10 mAb. The signal at the nuclear periphery with 21A10 mAb was much higher and the background lower than that with 2H10 and 13C2 antibosies.
|
|
|
| メーカー |
品番 |
包装 |
|
BAM
|
70-346
|
100 UG
|
※表示価格について
| 当社在庫 |
なし
|
| 納期目安 |
1週間程度
|
| 保存温度 |
-20℃
|
|