|
※サムネイル画像をクリックすると拡大画像が表示されます。
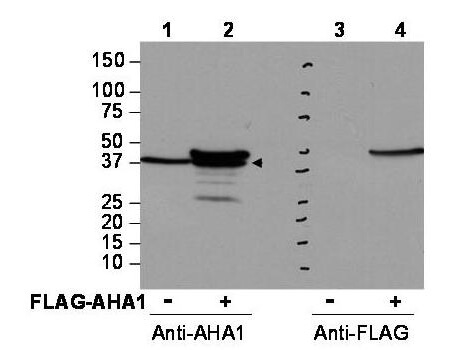
Western blot using Rockland's affinity purified anti-AHA1 antibody shows detection of AHA1 in Cos7 cells. For Lanes 2 and 4, Cos7 cells were transfected with pcDNA3-FLAG-AHA1. For Lanes 1 and 3, Cos7 cells were not transfected. Extracts (40 μg per lane) were electrophoresed and transferred to nitrocellulose. The membrane was probed with anti-AHA1 (lanes 1 and 2, 1:2,000 dilution) or anti-FLAG (lanes 3 and 4). The lower band seen in anti-AHA1 blotting (arrowhead) is endogenous AHA1. Personal Communication, Brad Scroggins, CCR-NCI, Bethesda, MD
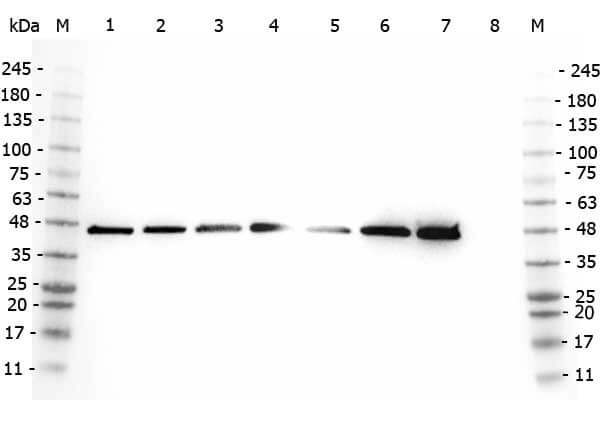
Western Blot of Rabbit anti-AHA1 antibody. Marker: Opal Pre-stained ladder (p/n MB-210-0500). Lane 1: HEK293 lysate (p/n W09-000-365). Lane 2: HeLa Lysate (p/n W09-000-364). Lane 3: MCF-7 Lysate (p/n W09-000-360). Lane 4: Jurkat Lysate (p/n W09-000-370). Lane 5: A431 Lysate (p/n W09-000-361). Lane 6: Raji Lysate (p/n W09-001-368). Lane 7: Ramos Lysate (p/n W09-000-GK4). Lane 8: NIH/3T3 Lysate (p/n W10-000-358). Load: 35 μg per lane. Primary antibody: AHA1 antibody at 1:2,000 for overnight at 4°C. Secondary antibody: Peroxidase rabbit secondary antibody (p/n 611-103-122) at 1:30,000 for 60 min at RT. Blocking Buffer: 1% Casein-TTBS (p/n MB-082) for 30 min at RT. Predicted/Observed size: 38 kDa, 38 kDa for AHA1.
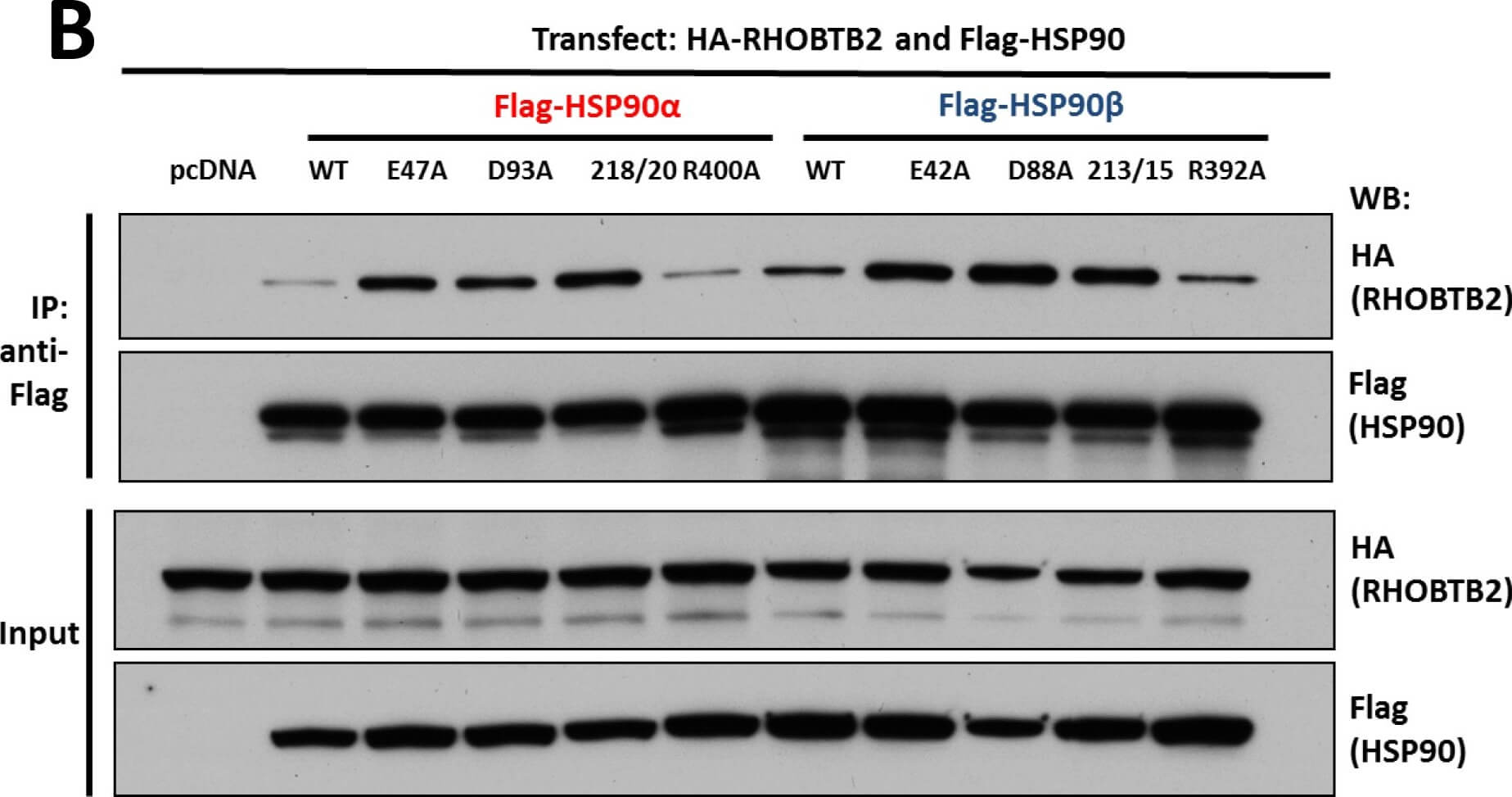
Interaction of KEAP1 and RHOBTB2 with HSP90 isoforms.(A) KEAP1interaction with HSP90 WT and mutants: HEK293 cells transfected with HA-KEAP1 and each FLAG-HSP90 construct were harvested, immunoprecipitated with anti-FLAG beads and western blotted for HA. Input lysates were normalized and run as controls. (B) RHOBTB2 interaction with HSP90 WT and mutants: HEK293 cells transfected with HA-RHOBTB2 and each FLAG-HSP90 construct were harvested, immunoprecipitated with anti-FLAG beads and western blotted for HA. Input lysates were normalized and run as controls. (C) Measurement of the relative interaction strength of KEAP1 and RHOBTB2 with each HSP90 isoform by LUMIER: HEK293 cells transfected with KEAP1 or RHOBTB2 and each HSP90 isoform were harvested, applied to a 96-well anti-FLAG plate and assayed for luciferase activity. The difference in relative interaction strength of HSP90α and HSP90β for KEAP1 and RHOBTB2 (each approximately 3-fold) was statistically significant (p<0.05) (see Methods). Figure provided by CiteAb. Source: PLoS One, PMID: 26517842.
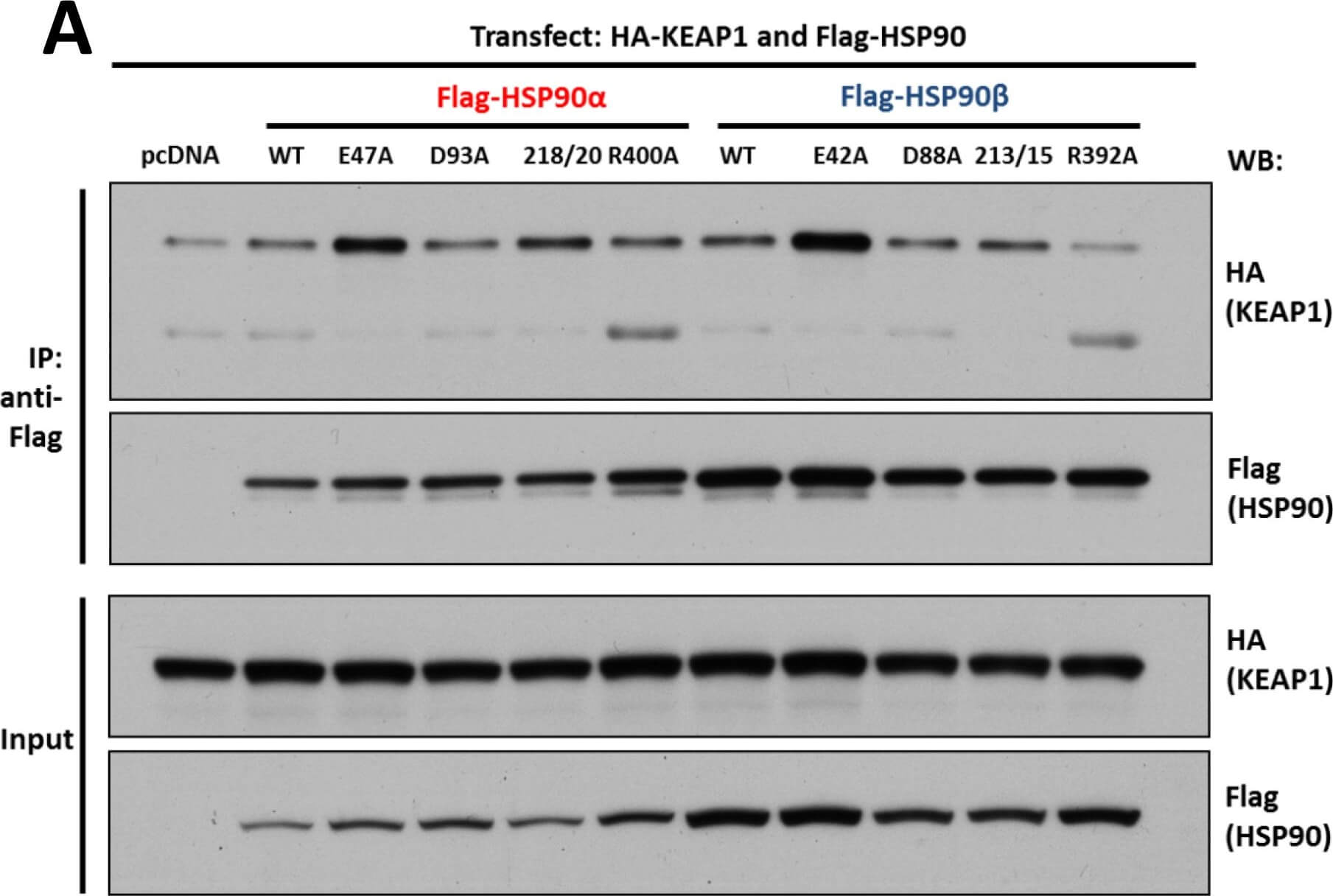
Interaction of KEAP1 and RHOBTB2 with HSP90 isoforms.(A) KEAP1interaction with HSP90 WT and mutants: HEK293 cells transfected with HA-KEAP1 and each FLAG-HSP90 construct were harvested, immunoprecipitated with anti-FLAG beads and western blotted for HA. Input lysates were normalized and run as controls. (B) RHOBTB2 interaction with HSP90 WT and mutants: HEK293 cells transfected with HA-RHOBTB2 and each FLAG-HSP90 construct were harvested, immunoprecipitated with anti-FLAG beads and western blotted for HA. Input lysates were normalized and run as controls. (C) Measurement of the relative interaction strength of KEAP1 and RHOBTB2 with each HSP90 isoform by LUMIER: HEK293 cells transfected with KEAP1 or RHOBTB2 and each HSP90 isoform were harvested, applied to a 96-well anti-FLAG plate and assayed for luciferase activity. The difference in relative interaction strength of HSP90α and HSP90β for KEAP1 and RHOBTB2 (each approximately 3-fold) was statistically significant (p<0.05) (see Methods). Figure provided by CiteAb. Source: PLoS One, PMID: 26517842.
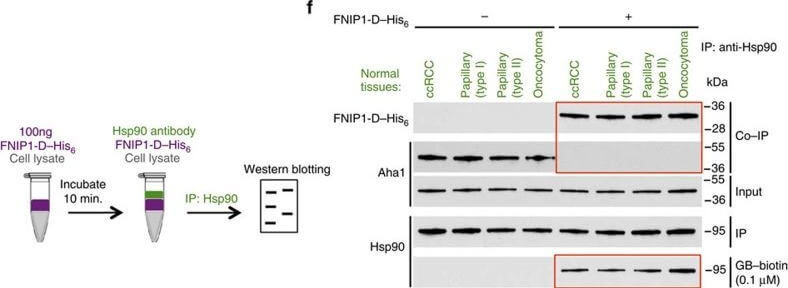
High levels of FNIPs make renal tumours sensitive to Hsp90 inhibitor GB.(a) Clear cell renal cell carcinoma (ccRCC), (b) Papillary type I, (c) Papillary type II, (d) Oncocytoma (Tumours, T) and adjacent normal tissues (Normal, N) were stained with haematoxylin and eosin (H&E). Proteins were also extracted from these tumours and adjacent normal tissues and incubated with indicated amounts of biotinylated GB followed by streptavidin agarose beads. Hsp90 was detected by immunoblotting. Expression of FNIP1 and FNIP2 in these samples was also detected by immunoblotting. (e) Hsp90 immunoprecipitated from tumours (T) and adjacent normal tissues (N) in a?d. Co-IP of FNIPs and Aha1 was examined by immunoblotting. (f) Lysates from normal tissues in a?d were incubated with or without 100?ng of pure FNIP1-D?His6 for 10?min. Hsp90 was immunoprecipitated and co-IP of FNIP1-D?His6 and Aha1 were shown by immunoblotting. Figure provided by CiteAb. Source: Nat Commun, PMID: 27353360.
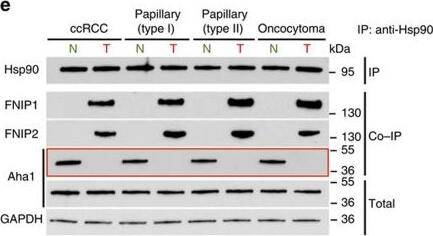
High levels of FNIPs make renal tumours sensitive to Hsp90 inhibitor GB.(a) Clear cell renal cell carcinoma (ccRCC), (b) Papillary type I, (c) Papillary type II, (d) Oncocytoma (Tumours, T) and adjacent normal tissues (Normal, N) were stained with haematoxylin and eosin (H&E). Proteins were also extracted from these tumours and adjacent normal tissues and incubated with indicated amounts of biotinylated GB followed by streptavidin agarose beads. Hsp90 was detected by immunoblotting. Expression of FNIP1 and FNIP2 in these samples was also detected by immunoblotting. (e) Hsp90 immunoprecipitated from tumours (T) and adjacent normal tissues (N) in a?d. Co-IP of FNIPs and Aha1 was examined by immunoblotting. (f) Lysates from normal tissues in a?d were incubated with or without 100?ng of pure FNIP1-D?His6 for 10?min. Hsp90 was immunoprecipitated and co-IP of FNIP1-D?His6 and Aha1 were shown by immunoblotting. Figure provided by CiteAb. Source: Nat Commun, PMID: 27353360.
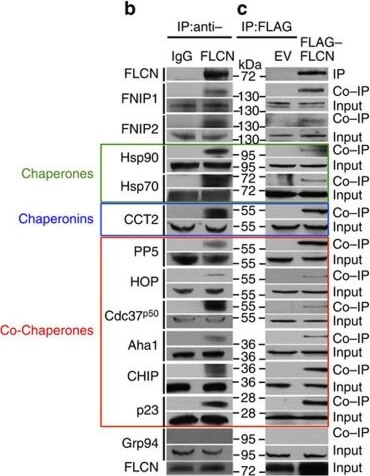
Folliculin is a new client of Hsp90.(a) FLAG?FLCN was expressed and isolated from HEK293 cells. Profile of interacting proteins determined by MALDI?time of flight. Red nodes represent chaperones and co-chaperones, blue nodes are chaperonins and green nodes are splicing factors and ribosomal proteins. (b) FLCN was isolated from HEK293 cell lysates using anti-FLCN or IgG (control) and immunoblotted with indicated antibodies to confirm protein interactions. (c) HEK293 cells were transiently transfected with FLAG?FLCN or empty vector control (EV), immunoprecipitated and immunoblotted with indicated antibodies to confirm interacting proteins. (d) HEK293 cells were treated with 10?μM of the Hsp70 inhibitor JG-98 at the indicated time points. FLCN protein stability in soluble and insoluble fraction was assessed by immunoblotting. (e) HEK293 cells were treated with 1?μM GB at the indicated time points. FLCN protein stability was assessed by immunoblotting. Akt and Phospho-S473-Akt were used as positive controls. (f) Hsp90α?FLAG was transiently expressed in HEK293 cells. Cells were treated with 1?μM GB for the indicated times. Hsp90α?FLAG was immunoprecipitated and co-IP of FLCN was examined by immunoblotting. (g) HEK293 cells were treated with 50?nM of the proteasome inhibitor bortezomib (BZ) for the indicated times. FLCN protein levels were evaluated at the indicated time points by immunoblotting (upper blots). HEK293 cells were also treated with 1?μM GB for 1?h before addition of 50?nM BZ. Immunoblotting was used to evaluate the FLCN level for the indicated time points (lower blots). (h) Empty vector (EV) or FLAG?FLCN was used to transiently transfect HEK293 cells for 24?h then treated for 4?h with either 50?nm BZ or 1?μM GB. FLAG?FLCN was immunoprecipitated and ubiquitination was examined by immunoblotting with a pan-anti-ubiquitin antibody. Figure provided by CiteAb. Source: Nat Commun, PMID: 27353360.
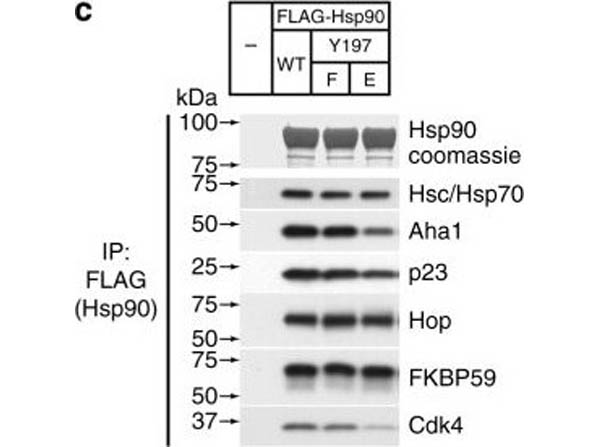
Assembly of complexes of Cdc37 and Hsp90 phosphomimetic variants with clients and cochaperones. a Binary complex formation between Cdc37 variants and bRaf (left), and between Hsp90β variants and Cdc37 (right), followed by ITC. The corresponding Kd values are displayed in the inset. Error bars in the Kd values correspond to the errors resulted in fitting of the data into a single binding site model. b HEK-293 cells were cotransfected with indicated HA-tagged Cdc37 and FLAG-tagged bRaf plasmids. After cell lysis, proteins were immunoprecipitated with anti-FLAG resin for 1?h at 4?°C with rotation. Bead pellets were washed and analyzed for Cdc37 interaction by SDS-PAGE/western blot, using anti-HA antibody. c HEK-293 cells were transfected with FLAG-tagged Hsp90, Hsp90Y197E, or Hsp90Y197F plasmids. After cell lysis, proteins were immunoprecipitated with anti-FLAG resin for 1?h at 4?°C with rotation. Bead pellets were washed three times before analysis by SDS-PAGE/western blot. Co-precipitating endogenous Hsp70, Aha1, p23, Hop, Fkbp59, and Cdk4 were detected with specific antibodies. d HEK-293 cells were transfected with the indicated Hsp90, androgen receptor (AR), and glucocorticoid receptor (GR) plasmids. Proteins were precipitated with GFP-Trap resin (left) or ANTI-FLAG M2 agarose (right) for 1?h at 4?°C with rotation. Bead pellets were washed three times with lysis buffer before analysis by SDS-PAGE/western blot as indicated. AR was visualized with anti-GFP antibody, GR was visualized with a specific antibody, and Hsp90 was visualized with anti-FLAG antibody Figure provided by CiteAb. Source: Nat Commun, PMID: 29343704.
|

|
|
Western blot using Rockland's affinity purified anti-AHA1 antibody shows detection of AHA1 in Cos7 cells. For Lanes 2 and 4, Cos7 cells were transfected with pcDNA3-FLAG-AHA1. For Lanes 1 and 3, Cos7 cells were not transfected. Extracts (40 μg per lane) were electrophoresed and transferred to nitrocellulose. The membrane was probed with anti-AHA1 (lanes 1 and 2, 1:2,000 dilution) or anti-FLAG (lanes 3 and 4). The lower band seen in anti-AHA1 blotting (arrowhead) is endogenous AHA1. Personal Communication, Brad Scroggins, CCR-NCI, Bethesda, MD
|
|
| 別品名 |
rabbit anti-AHA1 Antibody, Aha-1, Aha 1, Ahsa1 antibody, Activator of Hsp90 ATPase, Activator of 90 kDa heat shock protein ATPase homolog 1 antibody
|
| 交差種 |
Human
Monkey
|
| 適用 |
Western Blot
Enzyme Linked Immunosorbent Assay
|
| 免疫動物 |
Rabbit
|
| 標識物 |
Unlabeled
|
| 精製度 |
Affinity Purified
|
| GENE ID |
10598
|
| Accession No.(Gene/Protein) |
NP_001308370.1, O95433
|
| Gene Symbol |
AHSA1
|
| 参考文献 |
[Pub Med ID]26517842
|
|
| メーカー |
品番 |
包装 |
|
RKL
|
600-401-974
|
100 UG
|
※表示価格について
| 当社在庫 |
なし
|
| 納期目安 |
約10日
|
| 保存温度 |
-20℃
|
|








