|
※サムネイル画像をクリックすると拡大画像が表示されます。
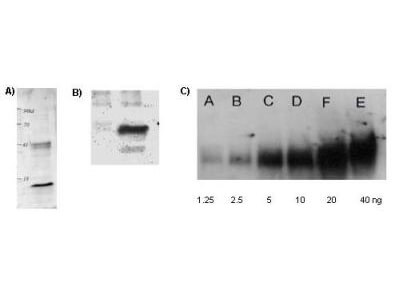
Rockland's Affinity Purified anti-Acetylated Lysine (AcK) antibody is shown to detect acetylated histone in TSA-treated mouse spleen cell lysate (Panel A); control (left lane) and TSA-treated mouse spleen cell lysate (right lane) in panel B; and in acetylated BSA loaded as indicated (panel C).
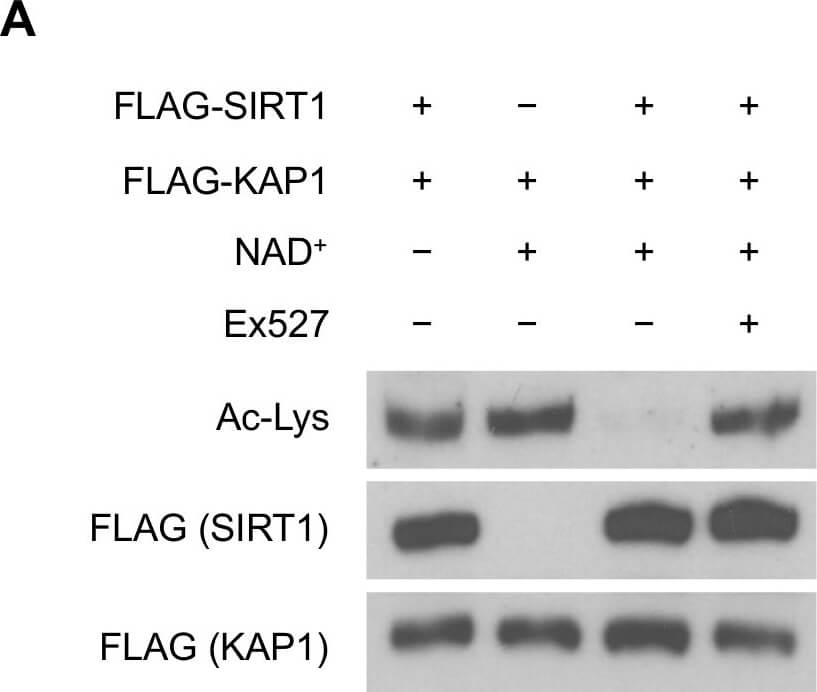
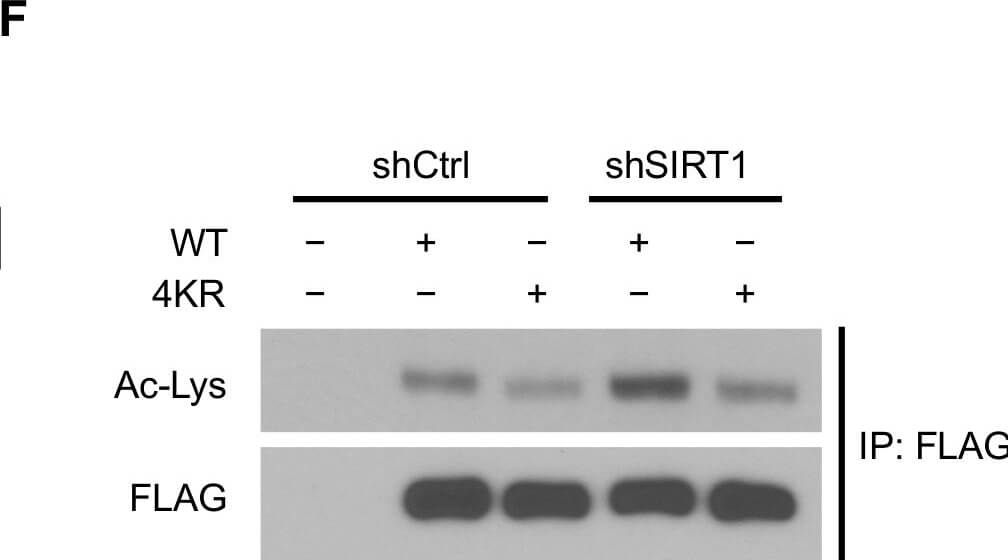
SIRT1 deacetylates KAP1 in vitro and in vivo.(A) SIRT1 deacetylates KAP1 in vitro. Exogenous FLAG-tagged KAP1 and SIRT1 were purified by anti-FLAG immunoprecipitation. Combination of purified proteins was incubated in deacetylation buffer supplemented with or without NAD+ cofactor. Ex527 (40μM) was added to block the deacetylase activity of SIRT1. (B) SIRT1 deacetylates KAP1 in vivo. FLAG-tagged KAP1 was transfected into control or SIRT1 depleted HEK293T cells. Cells were treated with or without IR 1 hour before harvest. Cell lysates were subjected to immunoprecipitation using anti-FLAG antibody, followed by western blot analysis to assess the total KAP1 acetylation level. Relative density of the overall acetyl lysine was quantified using ImageJ software. Acetylation level was normalized to corresponding FLAG band. (C) HEK293T cells transfected with FLAG-tagged KAP1 were treated with DMSO or Ex527 (20μM) for 6 hours before harvest. Cell lysates were then immunoprecipitated with FLAG-conjugated agarose beads, and the immunoprecipitates were blotted with anti-acetyl lysine antibody to determine the total acetylation level. (D and E) HEK293T cells were transfected with FLAG-tagged KAP1 and treated with DMSO or Ex527 (20μM) for 6 hours before harvest. Recombinant KAP1 was purified and sent for mass spectrometric analysis. Acetyl residues with >2-fold enhancement after inhibitor treatment were considered to be SIRT1 targeted sites. (F) Site-directed mutagenesis was applied to generate 4KR mutant. FLAG-tagged WT-KAP1 or 4KR mutant were transfected into control or SIRT1 depleted HEK293T cells. Total acetylation levels of recombinant WT-KAP1 and 4KR mutant were assessed by immunoblotting. Figure provided by CiteAb. Source: PLoS One, PMID: 25905708.
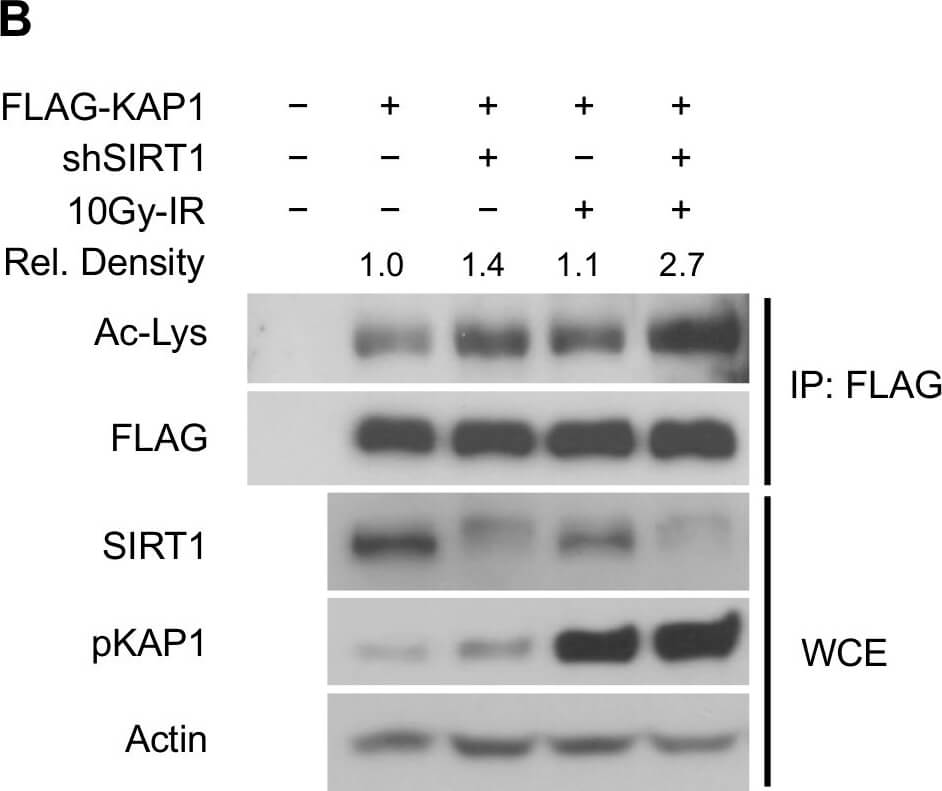
SIRT1 deacetylates KAP1 in vitro and in vivo.(A) SIRT1 deacetylates KAP1 in vitro. Exogenous FLAG-tagged KAP1 and SIRT1 were purified by anti-FLAG immunoprecipitation. Combination of purified proteins was incubated in deacetylation buffer supplemented with or without NAD+ cofactor. Ex527 (40μM) was added to block the deacetylase activity of SIRT1. (B) SIRT1 deacetylates KAP1 in vivo. FLAG-tagged KAP1 was transfected into control or SIRT1 depleted HEK293T cells. Cells were treated with or without IR 1 hour before harvest. Cell lysates were subjected to immunoprecipitation using anti-FLAG antibody, followed by western blot analysis to assess the total KAP1 acetylation level. Relative density of the overall acetyl lysine was quantified using ImageJ software. Acetylation level was normalized to corresponding FLAG band. (C) HEK293T cells transfected with FLAG-tagged KAP1 were treated with DMSO or Ex527 (20μM) for 6 hours before harvest. Cell lysates were then immunoprecipitated with FLAG-conjugated agarose beads, and the immunoprecipitates were blotted with anti-acetyl lysine antibody to determine the total acetylation level. (D and E) HEK293T cells were transfected with FLAG-tagged KAP1 and treated with DMSO or Ex527 (20μM) for 6 hours before harvest. Recombinant KAP1 was purified and sent for mass spectrometric analysis. Acetyl residues with >2-fold enhancement after inhibitor treatment were considered to be SIRT1 targeted sites. (F) Site-directed mutagenesis was applied to generate 4KR mutant. FLAG-tagged WT-KAP1 or 4KR mutant were transfected into control or SIRT1 depleted HEK293T cells. Total acetylation levels of recombinant WT-KAP1 and 4KR mutant were assessed by immunoblotting. Figure provided by CiteAb. Source: PLoS One, PMID: 25905708.
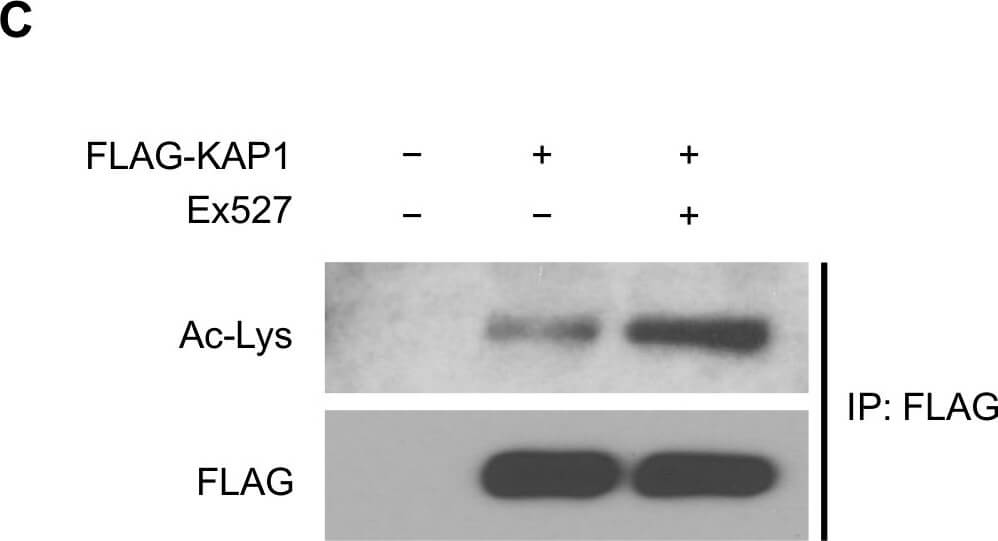
SIRT1 deacetylates KAP1 in vitro and in vivo.(A) SIRT1 deacetylates KAP1 in vitro. Exogenous FLAG-tagged KAP1 and SIRT1 were purified by anti-FLAG immunoprecipitation. Combination of purified proteins was incubated in deacetylation buffer supplemented with or without NAD+ cofactor. Ex527 (40μM) was added to block the deacetylase activity of SIRT1. (B) SIRT1 deacetylates KAP1 in vivo. FLAG-tagged KAP1 was transfected into control or SIRT1 depleted HEK293T cells. Cells were treated with or without IR 1 hour before harvest. Cell lysates were subjected to immunoprecipitation using anti-FLAG antibody, followed by western blot analysis to assess the total KAP1 acetylation level. Relative density of the overall acetyl lysine was quantified using ImageJ software. Acetylation level was normalized to corresponding FLAG band. (C) HEK293T cells transfected with FLAG-tagged KAP1 were treated with DMSO or Ex527 (20μM) for 6 hours before harvest. Cell lysates were then immunoprecipitated with FLAG-conjugated agarose beads, and the immunoprecipitates were blotted with anti-acetyl lysine antibody to determine the total acetylation level. (D and E) HEK293T cells were transfected with FLAG-tagged KAP1 and treated with DMSO or Ex527 (20μM) for 6 hours before harvest. Recombinant KAP1 was purified and sent for mass spectrometric analysis. Acetyl residues with >2-fold enhancement after inhibitor treatment were considered to be SIRT1 targeted sites. (F) Site-directed mutagenesis was applied to generate 4KR mutant. FLAG-tagged WT-KAP1 or 4KR mutant were transfected into control or SIRT1 depleted HEK293T cells. Total acetylation levels of recombinant WT-KAP1 and 4KR mutant were assessed by immunoblotting. Figure provided by CiteAb. Source: PLoS One, PMID: 25905708.
SIRT1 deacetylates KAP1 in vitro and in vivo.(A) SIRT1 deacetylates KAP1 in vitro. Exogenous FLAG-tagged KAP1 and SIRT1 were purified by anti-FLAG immunoprecipitation. Combination of purified proteins was incubated in deacetylation buffer supplemented with or without NAD+ cofactor. Ex527 (40μM) was added to block the deacetylase activity of SIRT1. (B) SIRT1 deacetylates KAP1 in vivo. FLAG-tagged KAP1 was transfected into control or SIRT1 depleted HEK293T cells. Cells were treated with or without IR 1 hour before harvest. Cell lysates were subjected to immunoprecipitation using anti-FLAG antibody, followed by western blot analysis to assess the total KAP1 acetylation level. Relative density of the overall acetyl lysine was quantified using ImageJ software. Acetylation level was normalized to corresponding FLAG band. (C) HEK293T cells transfected with FLAG-tagged KAP1 were treated with DMSO or Ex527 (20μM) for 6 hours before harvest. Cell lysates were then immunoprecipitated with FLAG-conjugated agarose beads, and the immunoprecipitates were blotted with anti-acetyl lysine antibody to determine the total acetylation level. (D and E) HEK293T cells were transfected with FLAG-tagged KAP1 and treated with DMSO or Ex527 (20μM) for 6 hours before harvest. Recombinant KAP1 was purified and sent for mass spectrometric analysis. Acetyl residues with >2-fold enhancement after inhibitor treatment were considered to be SIRT1 targeted sites. (F) Site-directed mutagenesis was applied to generate 4KR mutant. FLAG-tagged WT-KAP1 or 4KR mutant were transfected into control or SIRT1 depleted HEK293T cells. Total acetylation levels of recombinant WT-KAP1 and 4KR mutant were assessed by immunoblotting. Figure provided by CiteAb. Source: PLoS One, PMID: 25905708.
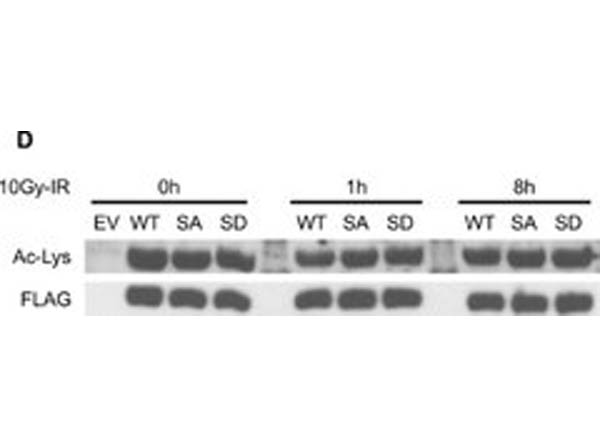
Acetylation does not crosstalk with IR-induced pSer824 and does not affect the co-repressor activity of KAP1.(A) Control and SIRT1 depleted HEK293T cells were harvested at indicated time points following 10Gy-IR. IR-induced KAP1 pSer824 was examined by phosphorylation specific antibody. (B) Cells transfected with FLAG-tagged WT-KAP1 or 4KR mutant were harvested at indicated time points following 4Gy-IR. Exogenous KAP1 was immunoprecipitated and pSer824 was examined by phosphorylation specific antibody. (C) Cyclin D3 luciferase construct was co-transfected with combinations of HA-tagged E2F1, FLAG-tagged KAP1, and 4KR mutant. Luciferase activity was measured 48 hours post transfection. (D) Cells transfected with FLAG-tagged WT-KAP1 or KAP1 phospho mutants (S824A and S824D) were harvested at indicated time points following 10Gy-IR. Recombinant KAP1 was purified by immunoprecipitation and the total acetylation level was assessed by immunoblotting. Figure provided by CiteAb. Source: PLoS One, PMID: 25905708.
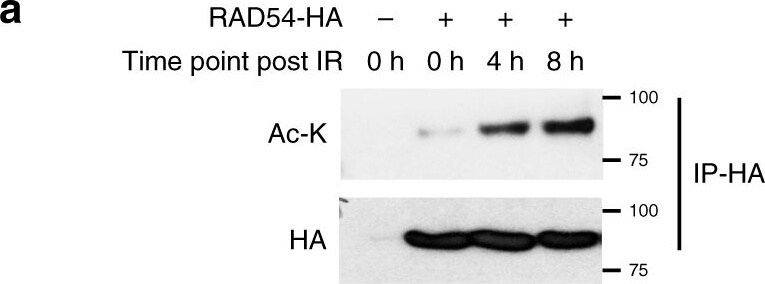
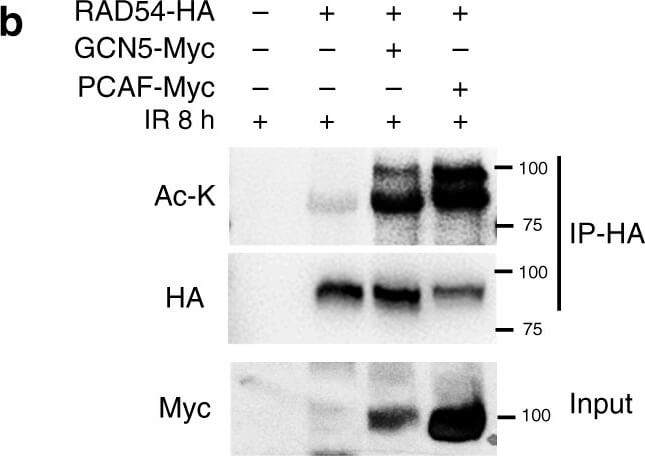
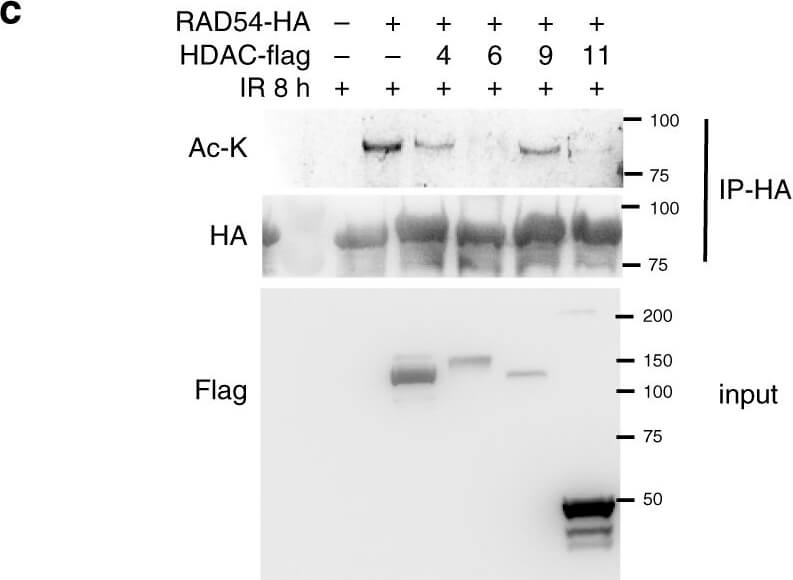
RAD54 acetylation is important for BRD9 recognition and HR activity.a?c RAD54 is acetylated by GCN5/PCAF and deacetylated by HDAC 6/HDAC11 following induction of DNA damage. a 293T cells were transfected with control or RAD54-HA plasmid. Twenty-four hours after transfection, cells were exposed to the 10-Gy IR and harvested at the indicated time points. Immunoprecipitation with anti-HA beads was performed. Blots were probed with the indicated antibodies. b 293T cells were transfected with the indicated plasmids. Twenty-four hours after transfection, cells were exposed to 10-Gy IR, and lysates were collected after 8?h. Immunoprecipitation with anti-HA beads was performed. Blots were probed with the indicated antibodies. c 293T cells were transfected with the indicated plasmids, treated as indicated, and subjected to immunoprecipitation as outlined in b. Blots were probed with the indicated antibodies. d, e RAD54 K515 acetylation is important for RAD51?RAD54 complex formation. d 293T cells were transfected with the indicated plasmids, treated as indicated, and subjected to immunoprecipitation as outlined in b. Blots were probed with the indicated antibodies. e 293T cells were transfected with the indicated plasmids, exposed to either no IR or 10-Gy IR as indicated, and subjected to immunoprecipitation as outlined in b. Blots were probed with the indicated antibodies. f?h RAD54 K515 acetylation is essential for HR activity. f, g U2OS cells were transfected with the indicated plasmids and exposed to 2-Gy IR. Cells were fixed after 8?h and stained for the indicated proteins. Representative immunofluorescence images of RAD51 (green) and RAD54 (red) are shown in f. Quantification of the indicated foci is shown in g. Representative data (mean?±?SEM) are shown from n?=?50 cells examined over three independent experiments. **p?
RAD54 acetylation is important for BRD9 recognition and HR activity.a?c RAD54 is acetylated by GCN5/PCAF and deacetylated by HDAC 6/HDAC11 following induction of DNA damage. a 293T cells were transfected with control or RAD54-HA plasmid. Twenty-four hours after transfection, cells were exposed to the 10-Gy IR and harvested at the indicated time points. Immunoprecipitation with anti-HA beads was performed. Blots were probed with the indicated antibodies. b 293T cells were transfected with the indicated plasmids. Twenty-four hours after transfection, cells were exposed to 10-Gy IR, and lysates were collected after 8?h. Immunoprecipitation with anti-HA beads was performed. Blots were probed with the indicated antibodies. c 293T cells were transfected with the indicated plasmids, treated as indicated, and subjected to immunoprecipitation as outlined in b. Blots were probed with the indicated antibodies. d, e RAD54 K515 acetylation is important for RAD51?RAD54 complex formation. d 293T cells were transfected with the indicated plasmids, treated as indicated, and subjected to immunoprecipitation as outlined in b. Blots were probed with the indicated antibodies. e 293T cells were transfected with the indicated plasmids, exposed to either no IR or 10-Gy IR as indicated, and subjected to immunoprecipitation as outlined in b. Blots were probed with the indicated antibodies. f?h RAD54 K515 acetylation is essential for HR activity. f, g U2OS cells were transfected with the indicated plasmids and exposed to 2-Gy IR. Cells were fixed after 8?h and stained for the indicated proteins. Representative immunofluorescence images of RAD51 (green) and RAD54 (red) are shown in f. Quantification of the indicated foci is shown in g. Representative data (mean?±?SEM) are shown from n?=?50 cells examined over three independent experiments. **p?
RAD54 acetylation is important for BRD9 recognition and HR activity.a?c RAD54 is acetylated by GCN5/PCAF and deacetylated by HDAC 6/HDAC11 following induction of DNA damage. a 293T cells were transfected with control or RAD54-HA plasmid. Twenty-four hours after transfection, cells were exposed to the 10-Gy IR and harvested at the indicated time points. Immunoprecipitation with anti-HA beads was performed. Blots were probed with the indicated antibodies. b 293T cells were transfected with the indicated plasmids. Twenty-four hours after transfection, cells were exposed to 10-Gy IR, and lysates were collected after 8?h. Immunoprecipitation with anti-HA beads was performed. Blots were probed with the indicated antibodies. c 293T cells were transfected with the indicated plasmids, treated as indicated, and subjected to immunoprecipitation as outlined in b. Blots were probed with the indicated antibodies. d, e RAD54 K515 acetylation is important for RAD51?RAD54 complex formation. d 293T cells were transfected with the indicated plasmids, treated as indicated, and subjected to immunoprecipitation as outlined in b. Blots were probed with the indicated antibodies. e 293T cells were transfected with the indicated plasmids, exposed to either no IR or 10-Gy IR as indicated, and subjected to immunoprecipitation as outlined in b. Blots were probed with the indicated antibodies. f?h RAD54 K515 acetylation is essential for HR activity. f, g U2OS cells were transfected with the indicated plasmids and exposed to 2-Gy IR. Cells were fixed after 8?h and stained for the indicated proteins. Representative immunofluorescence images of RAD51 (green) and RAD54 (red) are shown in f. Quantification of the indicated foci is shown in g. Representative data (mean?±?SEM) are shown from n?=?50 cells examined over three independent experiments. **p?
|

|
|
Rockland's Affinity Purified anti-Acetylated Lysine (AcK) antibody is shown to detect acetylated histone in TSA-treated mouse spleen cell lysate (Panel A); control (left lane) and TSA-treated mouse spleen cell lysate (right lane) in panel B; and in acetylated BSA loaded as indicated (panel C).
|
|
| 別品名 |
acetyl Lysine antibody, Acetylated lysine antibody, Lysine antibody
|
| 適用 |
Western Blot
|
| 免疫動物 |
Rabbit
|
| 標識物 |
Unlabeled
|
| 精製度 |
Affinity Purified
|
| 翻訳後修飾 |
アセチル化
|
| 参考文献 |
[Pub Med ID]32457312
|
| [注意事項] |
濃度はロットによって異なる可能性があります。メーカーDS及びCoAからご確認ください。
|
|
| メーカー |
品番 |
包装 |
|
RKL
|
600-401-939
|
100 UG
|
※表示価格について
| 当社在庫 |
なし
|
| 納期目安 |
約10日
|
| 保存温度 |
-20℃
|
|
※当社では商品情報の適切な管理に努めておりますが、表示される法規制情報は最新でない可能性があります。
また法規制情報の表示が無いものは、必ずしも法規制に非該当であることを示すものではありません。
商品のお届け前に最新の製品法規制情報をお求めの際はこちらへお問い合わせください。
|
※当社取り扱いの試薬・機器製品および受託サービス・創薬支援サービス(納品物、解析データ等)は、研究用としてのみ販売しております。
人や動物の医療用・臨床診断用・食品用としては、使用しないように、十分ご注意ください。
法規制欄に体外診断用医薬品と記載のものは除きます。
|
|
※リンク先での文献等のダウンロードに際しましては、掲載元の規約遵守をお願いします。
|
|
※CAS Registry Numbers have not been verified by CAS and may be inaccurate.
|









