| 抗原部位 |
a.a.95-108
|
| 標識物 |
Unlabeled
|
| 精製度 |
Affinity Purified
|
| 適用 |
Western Blot
Enzyme Linked Immunosorbent Assay
|
| 免疫動物 |
Rabbit
|
| Accession No.(Gene/Protein) |
55775699
|
| Tag情報 |
V5
|
| 形状 |
滅菌済み液状品
|
| 参考文献 |
[Pub Med ID]27600154, 29378775, 30323305, 21985091, 26304125, 31105869, 35014953
|
| [注意事項] |
濃度はロットによって異なる可能性があります。メーカーDS及びCoAからご確認ください。
|
|
※サムネイル画像をクリックすると拡大画像が表示されます。
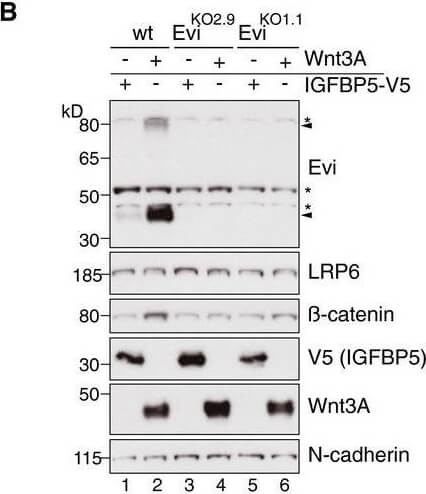
Wnt ligand production increases Evi protein levels In situ RNA hybridization (red dots) and immunohistochemistry (brown staining) of Evi were performed on sequential FFPE tissue slides of healthy colon and matched colon cancer tissue from five patients. The illustrated example is representative for three patients. Scale bar: 40?μm. Specificity of Evi probes was confirmed in Appendix?Fig S1B.Wild type (wt) or EviKO HEK293T cells were transfected with Wnt3A or IGFBP5 V5 expression plasmids and subjected to Western blot analysis. Specific Evi bands are indicated by arrows and unspecific bands by asterisks. Endogenous Evi is not only detectable as a monomeric form (46?kDa) but also as SDS resistant dimers (80?kDa). Clonal EviKO HEK293T cells were generated via CRISPR/Cas9 using Evi sgRNA #2 (EviKO2.9) or Evi sgRNA #1 (EviKO1.1; Appendix?Fig S2).HEK293T cells were transfected with Wnt expression plasmids and analyzed for endogenous Evi levels by immunoblotting with a mouse monoclonal Evi antibody (Biolegend, clone YJ5).HEK293T cells were transfected with the indicated overexpression constructs, treated with 100?ng/ml recombinant mouse Wnt3A (rec. W3A) or with 10?μM GSK3B inhibitor SB216763 for the indicated hours (h). The obtained cell lysates were used for Western blot analysis. Representative Western blots of three independent experiments are shown. B Actin or N cadherin were used as a loading control, and LRP6 served as a reference membrane protein. Scheme?showing that Evi is regulated through Wnt proteins within the Wnt producing cell. Canonical Wnt signaling can be activated by Wnt ligands, Dishevelled (Dvl) overexpression or by the GSK3B inhibitor SB216763.Source data are available online for this figure. Figure provided by CiteAb. Source: EMBO J, PMID: 29378775.
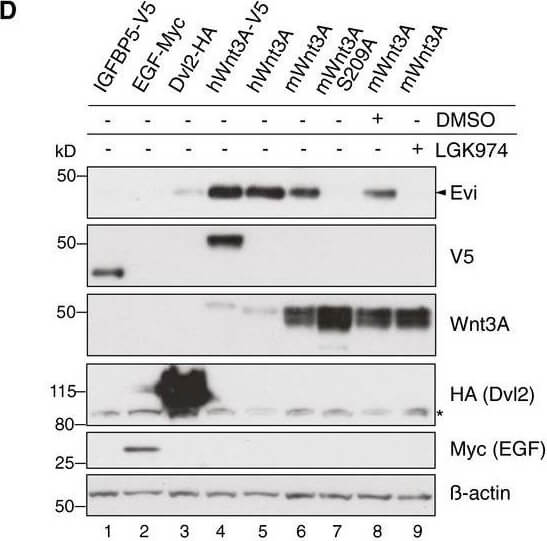
Evi stabilization is dependent on Wnt palmitoylation. Schematic illustration of the Porcn‐mediated Wnt palmitoylation, which is important for Evi‐Wnt interaction and which is blocked upon Porcn inhibition (LGK974), in PorcnKO cells and by using a palmitoylation‐deficient S209A Wnt3A mutant. Wild‐type or stable Wnt3‐and Wnt5B‐expressing HEK293T cells were treated with 5?μM LGK974 for 48?h and subjected to Western blot analysis. Western blot analysis of endogenous Evi in wt, PorcnKO, or EviKO HEK293T cells upon overexpression of Wnt3A or IGFBP5‐V5. PorcnKO1.2 and PorcnKO1.4 indicate clone #2 and clone #4 of PorcnKO HEK293T cells generated with Porcn sgRNA1 (Appendix?Fig S3). Clonal EviKO HEK293T cells were generated with Evi sgRNA2 (EviKO2.9; clone #9) or Evi sgRNA1 (EviKO1.1; clone #1; Appendix?Fig S2). Increase in total β‐catenin protein served as control for Wnt pathway activation. Western blot analysis of endogenous Evi in HEK293T cells transfected with the indicated overexpression plasmids. When indicated, the cells were additionally treated with 5?μM LGK974 for 48?h. Western blot analysis of endogenous Evi in HCT116 or A375 cells treated with 5?μM LGK974 or DMSO for the indicated hours (h). All Western blots are representative of three independent experiments. β‐Actin was used as a loading control, LRP6 as a reference membrane protein and EGF‐Myc and IGFBP5‐V5 as controls for secreted proteins. Specific Evi bands are indicated by arrows, and unspecific bands are marked by asterisks. Scheme: Wnt‐induced Evi stabilization is blocked in the absence of Wnt palmitoylation (PorcnKO, LGK974, Wnt3A S209A).Source data are available online for this figure. Figure provided by CiteAb. Source: EMBO J, PMID: 29378775.
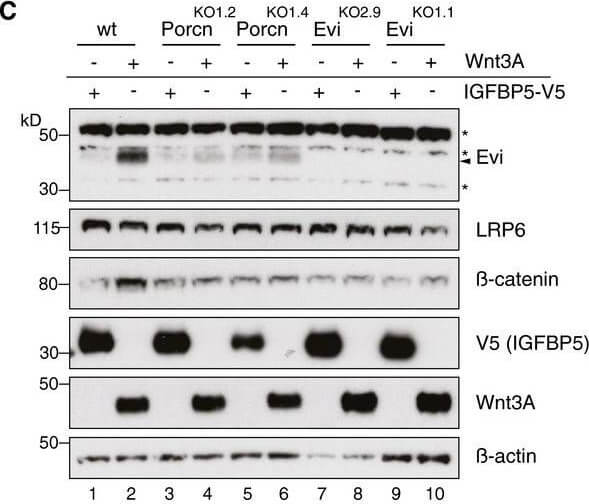
Evi stabilization is dependent on Wnt palmitoylation. Schematic illustration of the Porcn‐mediated Wnt palmitoylation, which is important for Evi‐Wnt interaction and which is blocked upon Porcn inhibition (LGK974), in PorcnKO cells and by using a palmitoylation‐deficient S209A Wnt3A mutant. Wild‐type or stable Wnt3‐and Wnt5B‐expressing HEK293T cells were treated with 5?μM LGK974 for 48?h and subjected to Western blot analysis. Western blot analysis of endogenous Evi in wt, PorcnKO, or EviKO HEK293T cells upon overexpression of Wnt3A or IGFBP5‐V5. PorcnKO1.2 and PorcnKO1.4 indicate clone #2 and clone #4 of PorcnKO HEK293T cells generated with Porcn sgRNA1 (Appendix?Fig S3). Clonal EviKO HEK293T cells were generated with Evi sgRNA2 (EviKO2.9; clone #9) or Evi sgRNA1 (EviKO1.1; clone #1; Appendix?Fig S2). Increase in total β‐catenin protein served as control for Wnt pathway activation. Western blot analysis of endogenous Evi in HEK293T cells transfected with the indicated overexpression plasmids. When indicated, the cells were additionally treated with 5?μM LGK974 for 48?h. Western blot analysis of endogenous Evi in HCT116 or A375 cells treated with 5?μM LGK974 or DMSO for the indicated hours (h). All Western blots are representative of three independent experiments. β‐Actin was used as a loading control, LRP6 as a reference membrane protein and EGF‐Myc and IGFBP5‐V5 as controls for secreted proteins. Specific Evi bands are indicated by arrows, and unspecific bands are marked by asterisks. Scheme: Wnt‐induced Evi stabilization is blocked in the absence of Wnt palmitoylation (PorcnKO, LGK974, Wnt3A S209A).Source data are available online for this figure. Figure provided by CiteAb. Source: EMBO J, PMID: 29378775.
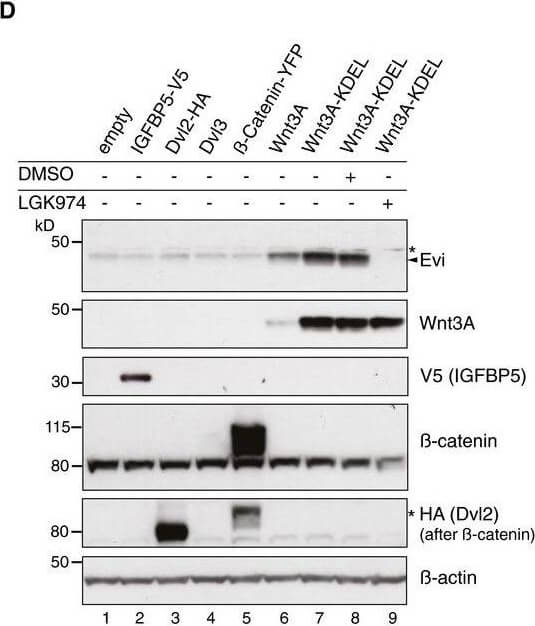
Evi poly‐ubiquitination is regulated by the presence of Wnt proteinsHEK293T cells were transfected with Wnt3A or IGFBP5‐V5 expression constructs and treated with MG132 at the indicated concentrations for 24?h. Cell lysates were analyzed for endogenous Evi by immunoblotting. Total β‐catenin protein was used to assess MG132 efficiency. Wild‐type (wt), stable Wnt3‐expressing, or EviKO2.9 HEK293T cells were treated with 1?μM MG132 for 24?h. TUBE2 immunoprecipitates were assayed for endogenous Evi or K48 poly‐ubiquitin by immunoblotting. To confirm specificity of the TUBE2 assay, Ctrl agarose beads were used as control. The asterisk at the β‐actin blot indicates Wnt3A proteins blotted before membrane stripping. Scheme?illustrating ubiquitination and proteasomal degradation of Evi, which is blocked in the presence of Wnt ligands.HEK293T cells were transfected with the indicated plasmids and additionally treated with 5?μM LGK974 for 48?h when indicated. In case of Wnt3A‐KDEL, the ER‐retaining sequence KDEL was C‐terminally fused to Wnt3A. Dvl2‐HA, Dvl3, and β‐catenin‐YFP overexpression was used as negative control to verify that Evi stabilization was not due to downstream activation of Wnt signaling. All Western blots are representative of three independent experiments. β‐Actin was used as loading control. Specific Evi bands are marked by arrows and unspecific bands by asterisks. Source data are available online for this figure. Figure provided by CiteAb. Source: EMBO J, PMID: 29378775.
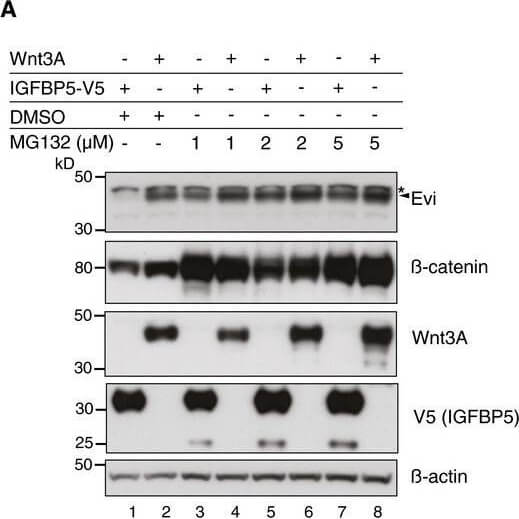
Evi poly‐ubiquitination is regulated by the presence of Wnt proteinsHEK293T cells were transfected with Wnt3A or IGFBP5‐V5 expression constructs and treated with MG132 at the indicated concentrations for 24?h. Cell lysates were analyzed for endogenous Evi by immunoblotting. Total β‐catenin protein was used to assess MG132 efficiency. Wild‐type (wt), stable Wnt3‐expressing, or EviKO2.9 HEK293T cells were treated with 1?μM MG132 for 24?h. TUBE2 immunoprecipitates were assayed for endogenous Evi or K48 poly‐ubiquitin by immunoblotting. To confirm specificity of the TUBE2 assay, Ctrl agarose beads were used as control. The asterisk at the β‐actin blot indicates Wnt3A proteins blotted before membrane stripping. Scheme?illustrating ubiquitination and proteasomal degradation of Evi, which is blocked in the presence of Wnt ligands.HEK293T cells were transfected with the indicated plasmids and additionally treated with 5?μM LGK974 for 48?h when indicated. In case of Wnt3A‐KDEL, the ER‐retaining sequence KDEL was C‐terminally fused to Wnt3A. Dvl2‐HA, Dvl3, and β‐catenin‐YFP overexpression was used as negative control to verify that Evi stabilization was not due to downstream activation of Wnt signaling. All Western blots are representative of three independent experiments. β‐Actin was used as loading control. Specific Evi bands are marked by arrows and unspecific bands by asterisks. Source data are available online for this figure. Figure provided by CiteAb. Source: EMBO J, PMID: 29378775.
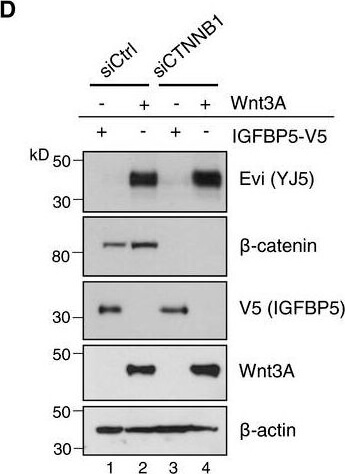
Evi is not transcriptionally regulated by WntA, BFPKM‐normalized RNA‐seq. data of the TCGA Research Network (TCGA‐COAD; http://cancergenome.nih.gov/; 09/25/2017) were log‐transformed to illustrate the relative expression of (A) Wnt3 and (B) Evi in healthy colon (41) versus colon adenocarcinoma (456). The distribution into tumor and healthy samples was based on their barcodes as described in TCGA Wiki. Statistical significance of gene expression differences was determined using a Student's t‐test under the alternative hypothesis H1 that gene expression is higher in tumors compared to healthy tissue. The boxplot diagram shows the median as line within the box, the 25th and 75th percentiles as the upper and lower part of the box, the 10th and 90th percentiles as error bars and outliers as circles.CHEK293T cells were transfected with the indicated expression constructs, treated with 100?ng/ml recombinant mouse Wnt3A (rec. W3A) or with 10?μM GSK3 inhibitor SB216763 for the indicated hours (h). AXIN2 and Evi mRNA levels were analyzed by qRTPCR and normalized to GAPDH expression. Results are shown as mean?±?s.d. from three independent experiments. D, D′Twenty‐four hours after reverse transfection with Ctrl or CTNNB1 siRNA, HEK293T cells were transfected with Wnt3A or IGFBP5‐V5 expression plasmids and analyzed (D) for the indicated proteins via immunoblotting or (D′) for canonical Wnt activity using the TCF‐Luciferase Wnt reporter assay. Immunoblotting is representative of three independent experiments, and Wnt reporter activity was calculated as mean from three independent experiments?±?s.d.Source data are available online for this figure. Figure provided by CiteAb. Source: EMBO J, PMID: 29378775.
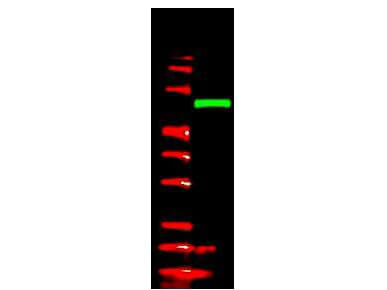
Anti-V5 epitope tag polyclonal antibody detects V5-tagged recombinant protein by western blot. This antibody was used at 1.0 ug/ml to detect 0.05 ug (lane 2) of full-length recombinant mouse serum albumin containing the V5 epitope tag at the carboxy end. Comparison to MW markers (lane 1) indicates detection of monomeric V5 tagged albumin. A 4-20% gradient gel was used to separate the protein by SDS-PAGE under non-reducing conditions. The protein was transferred to nitrocellulose using standard methods. After blocking the membrane was probed with the primary antibody overnight at 4 C followed by washes and reaction with a 1:10,000 dilution of IRDye 800 conjugated Gt-a-Rabbit IgG [H&L] (code 611-132-122) for 45 min at room temperature. LICOR's OdysseyR Infrared Imaging System was used to scan and process the image. Other detection systems will yield similar results.
|

|
|
Wnt ligand production increases Evi protein levels In situ RNA hybridization (red dots) and immunohistochemistry (brown staining) of Evi were performed on sequential FFPE tissue slides of healthy colon and matched colon cancer tissue from five patients. The illustrated example is representative for three patients. Scale bar: 40?μm. Specificity of Evi probes was confirmed in Appendix?Fig S1B.Wild type (wt) or EviKO HEK293T cells were transfected with Wnt3A or IGFBP5 V5 expression plasmids and subjected to Western blot analysis. Specific Evi bands are indicated by arrows and unspecific bands by asterisks. Endogenous Evi is not only detectable as a monomeric form (46?kDa) but also as SDS resistant dimers (80?kDa). Clonal EviKO HEK293T cells were generated via CRISPR/Cas9 using Evi sgRNA #2 (EviKO2.9) or Evi sgRNA #1 (EviKO1.1; Appendix?Fig S2).HEK293T cells were transfected with Wnt expression plasmids and analyzed for endogenous Evi levels by immunoblotting with a mouse monoclonal Evi antibody (Biolegend, clone YJ5).HEK293T cells were transfected with the indicated overexpression constructs, treated with 100?ng/ml recombinant mouse Wnt3A (rec. W3A) or with 10?μM GSK3B inhibitor SB216763 for the indicated hours (h). The obtained cell lysates were used for Western blot analysis. Representative Western blots of three independent experiments are shown. B Actin or N cadherin were used as a loading control, and LRP6 served as a reference membrane protein. Scheme?showing that Evi is regulated through Wnt proteins within the Wnt producing cell. Canonical Wnt signaling can be activated by Wnt ligands, Dishevelled (Dvl) overexpression or by the GSK3B inhibitor SB216763.Source data are available online for this figure. Figure provided by CiteAb. Source: EMBO J, PMID: 29378775.
|
|
|
| メーカー |
品番 |
包装 |
|
RKL
|
600-401-378
|
100 UG
|
※表示価格について
| 当社在庫 |
なし
|
| 納期目安 |
約10日
|
| 保存温度 |
-20℃
|
|