| 別品名 |
Daunomycin, Leukaemomycin C, Rubidomycin, RP-13057, Cerubidin., (8S-cis)-8-Acetyl-10-[(3-amino-2,3,6-trideoxy-α-L- lyxo-hexopyranosyl)oxy]-7,8,9,10-tetrahydro-6,8,11- trihydroxy-1-methoxy-5,12-naphthacenedione
|
| 別包装 |
あり
|
| 純度 |
>=98%
|
| 分子量 |
564
|
| CAS RN® |
23541-50-6
|
| 化合物の概要 |
Anthracycline, DNA intercalator; topoisomerase II inhibitor.
|
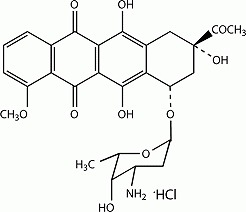
| 構造 |
C27H29NO10HCl
|
| 溶媒 |
Soluble in water or ethanol.
|
| 使用目的 |
Daunorubicin is an anthracycline first produced by Streptomyces. Daunorubicin exhibits anticancer chemotherapeutic activity and is clinically used to treat various cancers, including neuroblastoma and acute myelogenous leukemia (AML). Like other anthracyclines, daunorubicin acts as an intercalator and inhibitor of topoisomerase II. Daunorubicin also promotes histone H2AX eviction from chromatin, inhibiting DNA repair mechanisms.
|
| その他 |
[融点]188℃-190℃(dec.)
[外観]Orange-red Crystalline Powder
|
| 参考文献 |
Pang B, Qiao X, Janssen L, et al. Drug-induced histone eviction from open chromatin contributes to the chemotherapeutic effects of doxorubicin. Nat Commun. 2013;4:1908. PMID: 23715267.Quigley GJ, Wang AH, Ughetto G, et al. Molecular structure of an anticancer drug-DNA complex: daunomycin plus d(CpGpTpApCpG). Proc Natl Acad Sci U S A. 1980 Dec;77(12):7204-8. PMID: 6938965.
|
|
※サムネイル画像をクリックすると拡大画像が表示されます。
|
|
|
|
| メーカー |
品番 |
包装 |
|
LKT
|
D0182
|
100 MG
|
※表示価格について
| 当社在庫 |
なし
|
| 納期目安 |
1週間程度
|
| 保存温度 |
4℃
|
|